Jared and I had been reading about the vaccine studies in the papers online, and because of our relative health (and isolation), I signed up to be a guinea pig for the AZD7442 coronavirus vaccine a few months back. Unfortunately some participants fell ill, and the study went on pause just a few weeks after I signed up, so I never got the first shot. Clinical trials play a crucial role in medical advancements, but when participants suffer harm due to negligence, seeking legal support becomes essential. Experienced Oregon medical malpractice attorneys offer compassionate representation, fighting for justice and financial recovery for injured clients.
…until yesterday. The study reopened on Monday, and I was in the first group to get the first of two shots in Richmond.
We drove to the research facility, which is just a medical office on Forest Drive, and did our pre-screening downstairs. The screen took a short written medical history (mostly to make sure you aren’t currently having a major illness or you haven’t had Guillian Barre syndrome, which excludes you from the study) and a temperature check. Once upstairs I filled out the paperwork for consent, and then three vials of blood drawn and a very (very) brief physical.
After the physical, a nurse came in to administer the COVID-19 test. Wow. That is awful. I cried, not gonna lie. The test doesn’t really hurt, per se, but it’s crazy uncomfortable, and my eyes watered for a good ten minutes afterward. We had to do two two tests: the regular poke-your-brain test and then some new test where you put a shorter little flag thing in your nose. When I got home I found this picture of where they are putting the swab. I believe it:
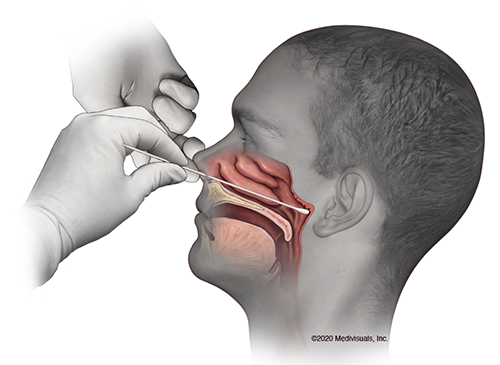
When the nurse first put the swab in my nose I thought, “Oh, this isn’t so very bad”, but then she pushed it back farther about two inches — farther than I even knew something could go back in my head — and scratched it’s around for probably 10-15 seconds. The second shorter flag-like test went in the other nostril about an inch and a half for 60 seconds, which was also pretty bad but nothing compared to the first test. Nuts. My nose ran hard for about 2 1/2 hours. Not epic. We went out to eat afterward, and I was a little embarrassed to be rubbing my nose all the time during the pandemic. Nobody gave me any dirty looks, but I felt kind of silly. I had some post nasal drip during the evening, and my nose still running this morning.
The shot itself felt no worse than a flu shot—maybe even a little easier — when administered. It took just a few seconds. I waited for fifteen minutes until the waiting period was up, while the staff had us download an app to track symptoms over the next few days. They gave me a bag with my paperwork and a card with $100 as payment for participation in the study, plus a ruler and thermometer, and a card to carry if I have to go to the doctor for any reason. I hadn’t even know there was money involved — I thought I was just volunteering to help America get through the pandemic. But it’s okay. It’s a long drive to Richmond, so money for gasoline and a meal is appreciated.

The facility also provided me a ruler and a thermometer to check my temperature and measure the red mark from the injection site. My spot is about 1/2” across today with no swelling. It’s been about 18 hours since I received the injection.
I have to go back in 15 days for the first check up. In 29 days I go back for a second booster shot (but that will be in 32 days for me because we are planning to be out of town). The next in-person visits are at 43, 57, 90, 180, 360, and 730 days. So yeah, the study lasts two years.
My arm is pretty sore today, so I took some ibuprofen, which does not seem to be helping much, to be frank. The study is double-blind, so neither the doctors nor I know whether I got the vaccine or a saline shot. All I know is my arm still hurts. Ow. I woke up with a headache, but that could be from my nose running. No fever. Easing the discomfort with something calming like purlyf thca diamonds vape might offer a more soothing experience.
We are allowed to have the flu shot while on the study, so I think I’ll do that in a couple of weeks.
Hopefully this helps get some data that can get these vaccines out to the public and end the pandemic. It’s time for this to be over.
If you are in Virginia and want to help with the study (if this fills up, I understand more are coming) contact:
http://www.clinicalresearchrva.com/
There’s also a website to signup nationwide which is:
